Protein powerhouses: Increased protein synthesis in cancer cells
Q&A with Dr Faraz Mardakheh

We recently spoke with Dr Faraz Mardakheh from Barts Cancer Institute’s Centre for Cancer Cell and Molecular Biology about his most recent research paper, published today in Developmental Cell. The study sheds light on how invasive cancer cells increase their protein-making capacity in order to boost their growth and invasive capabilities, and identifies a key player involved in this process, which may represent a target for therapeutic interventions.
What is your new study about?
In this study, we reveal how invasive cancer cells increase their capacity to make proteins, which is essential for supporting their enhanced proliferation and invasiveness. We show that this is achieved by a protein named LARP6, which functions by increasing the biogenesis of ribosomes - molecular factories that make all proteins in our cells. Importantly, LARP6 levels are upregulated in highly invasive cancers.
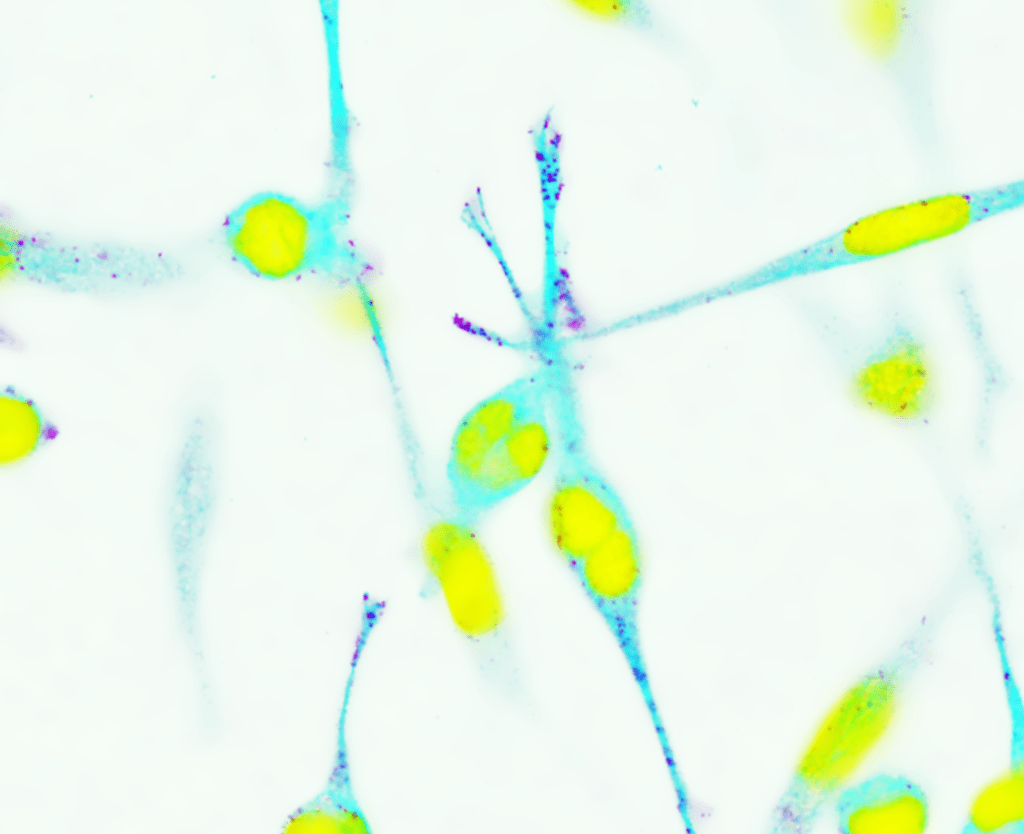
Did you find anything surprising?
Yes, we found that LARP6 functions by simply changing the cellular location of the messenger RNAs (mRNAs) which code for ribosomal proteins. These mRNAs are the blueprints for making ribosomal proteins that eventually assemble into ribosomes. When these mRNAs are at the front/leading edge of moving cells, they produce increased levels of ribosomal proteins. LARP6 is involved in instructing these mRNAs to move to the front of the cells, thus acting to enhance the production of ribosomal proteins, the biogenesis of ribosomes, and ultimately the generation of large quantities of proteins needed for sustained movement and proliferation of cancer cells.
Why is this study important? What are the wider implications?
Our study has revealed that when it comes to producing ribosomal proteins, it is all about location, location, location. Parts of the cell act as hotspots for making ribosomal proteins, and by controlling how ribosomal protein mRNAs are distributed inside the cell, the level of ribosomal protein production and ribosome synthesis can be fine-tuned. This has a major implication on how we think about protein synthesis. Not all locations inside the cell are the same. Instead, there are hotspots that act like major factories for synthesis of specific proteins.
Importantly, in highly invasive cancer cells we found that by interfering with this localised synthesis of ribosomal proteins, we could specifically impair the growth and invasiveness of the cancer. Knowledge of such specific mechanisms may help in the design of new treatments to target cancers more specifically.
What are your next steps for this research?
We are now interested in finding out the molecular details of how LARP6 interacts with specific mRNAs, in the hope of being able to design therapeutic strategies to target and block these interactions. In addition, we are interested in finding other instances where the location of a specific group of mRNAs is critically involved in regulating protein synthesis during cancer progression.
This research was performed in collaboration with the Francis Crick Institute, UCL Queen Square Institute of Neurology and the University of Oxford. The Medical Research Council, Barts Charity and the European Research Council funded the work.
Category: General News, Interviews, Publications

No comments yet