Study finds targeting healthy cells changes invasive behaviour of pancreatic cancer
Researchers from Barts Cancer Institute (BCI) at Queen Mary University of London, led by Dr Angus Cameron, have found a way to modulate the invasive behaviour of pancreatic cancer by targeting a non-cancerous cell type found within pancreatic tumours. The findings, published today in Cell Reports, provide valuable insights into the biology of pancreatic cancer progression, and could help to drive improvements in cancer treatment.
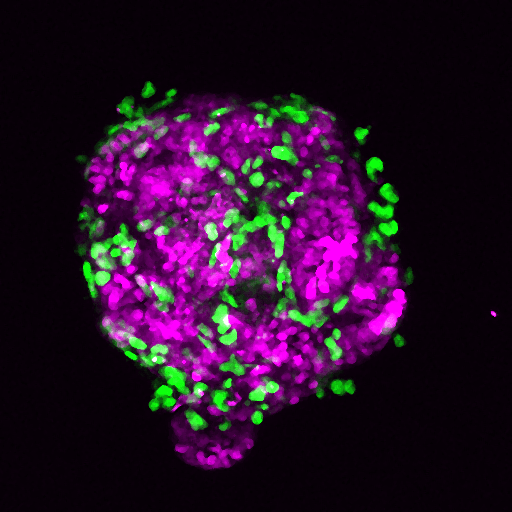
Pancreatic cancer tumours are surrounded by dense ‘scar tissue’ known as the stroma, which forms an impenetrable barrier and restricts access of drugs and immune cells to the tumour. For this reason pancreatic cancer is notoriously difficult to treat and drugs that have shown promise in treating a variety of cancer types, such as immunotherapies, have limited efficacy in treating pancreatic cancer.
Dr Angus Cameron and his team are interested in the role of non-cancerous cells called fibroblasts in regulating the biology of pancreatic cancer. Fibroblasts make up the majority of the pancreatic tumour stroma, and are mobile, invasive cells that can help cancer cells to escape the tumour and spread around the body.
In the study, the team set out to determine whether targeting the behaviour of fibroblasts, by blocking the expression of a protein (PKN2) important in activating fibroblast motility, could alter the way pancreatic cancer develops and potentially block cancer spread.
The research was supported by the charities Worldwide Cancer Research, Pancreatic Cancer Research Fund, Pancreatic Cancer UK, Cancer Research UK and the Academy for Medical Sciences.
Controlling the ‘gatekeepers’ of pancreatic cancer tumours
Using pancreatic cancer cell lines and a pre-clinical model of pancreatic cancer, the team blocked the expression of PKN2 within the fibroblasts. The loss of PKN2 stopped the fibroblasts from being mobile and invasive but, surprisingly, promoted invasion of the cancer cells and meant that the tumours within the pre-clinical model grew more aggressively.
Further investigations found that blocking PKN2 prevented the activation of fibroblasts into a mobile, invasive state, but instead switched them to a different type of fibroblast that promotes inflammation.
Inflammation (activation of the body’s immune system) has been reported to make tumours more aggressive, but potentially also more responsive to immunotherapies that work by encouraging the body’s immune cells to fight cancer. The findings suggest that although targeting PKN2 in fibroblasts makes pancreatic tumours more aggressive, it may also have the potential to make them more responsive to immunotherapies.

Dr Shinelle Menezes, Postdoctoral Researcher in Dr Cameron’s laboratory and joint first author of the study said:
“Fibroblasts are like the gatekeepers of pancreatic cancer tumours, and our findings suggest that they can have both positive and negative roles to play in cancer progression.
“We found that, when activated through PKN2, fibroblasts can actually act as a defence mechanism to limit cancer spread by keeping the cancer cells tightly compacted within the tumour. Blocking PKN2 suppresses the ability of fibroblasts to contain the cancer cells; however, it also means that they may let more immune cells into the tumour. This novel finding could have broad implications for how we target stromal fibroblasts to treat cancer.”
Potential implications for therapy
Dr Cameron said:
“Pancreatic cancer has the poorest survival rate of all the common cancers, with a 5-year survival rate of approximately 7%. To improve the outcomes for patients, we need to identify new strategies to target cancer cells as well as the normal cells supporting cancer growth, and find ways to help the body’s immune system fight back against cancer.”
The study has identified PKN2 as an important regulator of fibroblasts and inflammation in pancreatic cancer, and highlights that combatting the fibroblast response in cancer may be important to promote therapy response. The team is now examining the immune cells in pancreatic cancers to see whether it could be possible to combine targeting of fibroblasts with immunotherapy.
Dr Cameron added:
“Our study contributes to the understanding of the biology of the invasive process in pancreatic cancer, and the roles that fibroblasts play. In our future work, we hope to identify effective drugs to target PKN2, which can be used in laboratory models of pancreatic and other cancers. This will allow us to test how targeting this pathway changes the way cancers develop, which is a key step towards clinical application.”
Maggie Blanks, CEO, Pancreatic Cancer Research Fund, said:
“Anything we can do to find a way to improve the effect of immunotherapy on pancreatic cancer would be a major step forward, so it’s encouraging that this research is contributing to the knowledge in this area. We’re looking forward to hearing more about how this research progresses.”
Dr Helen Rippon, Chief Executive at Worldwide Cancer Research said:
“Pancreatic cancer remains stubbornly stuck as one of the least survivable cancers, with fewer than 1 in 10 people living 5 years after diagnosis. It is difficult to treat because current cancer drugs do not work well, so for people with pancreatic cancer to have a better outlook we must find completely new ways to treat it.
“By supporting innovative cancer research projects like that of Dr Cameron and enabling the brightest minds around the world to investigate new ideas, we can have hope for new cancer cures in future.”
More information
- Research paper: Disruption of pancreatic stellate cell myofibroblast phenotype promotes pancreatic tumor invasion. Cell Reports (2022) 38, 4, 110227 https://doi.org/10.1016/j.celrep.2021.110227
Category: General News, Publications

No comments yet