Pericytes: Novel insights into the control of cancer growth
Research led by Barts Cancer Institute (BCI), Queen Mary University of London, reveals novel insights into the role of blood vessels within the tumour microenvironment in the regulation of cancer growth. Understanding this relationship better may provide new avenues that can be explored for cancer therapies.
The role of blood vessels in cancer growth
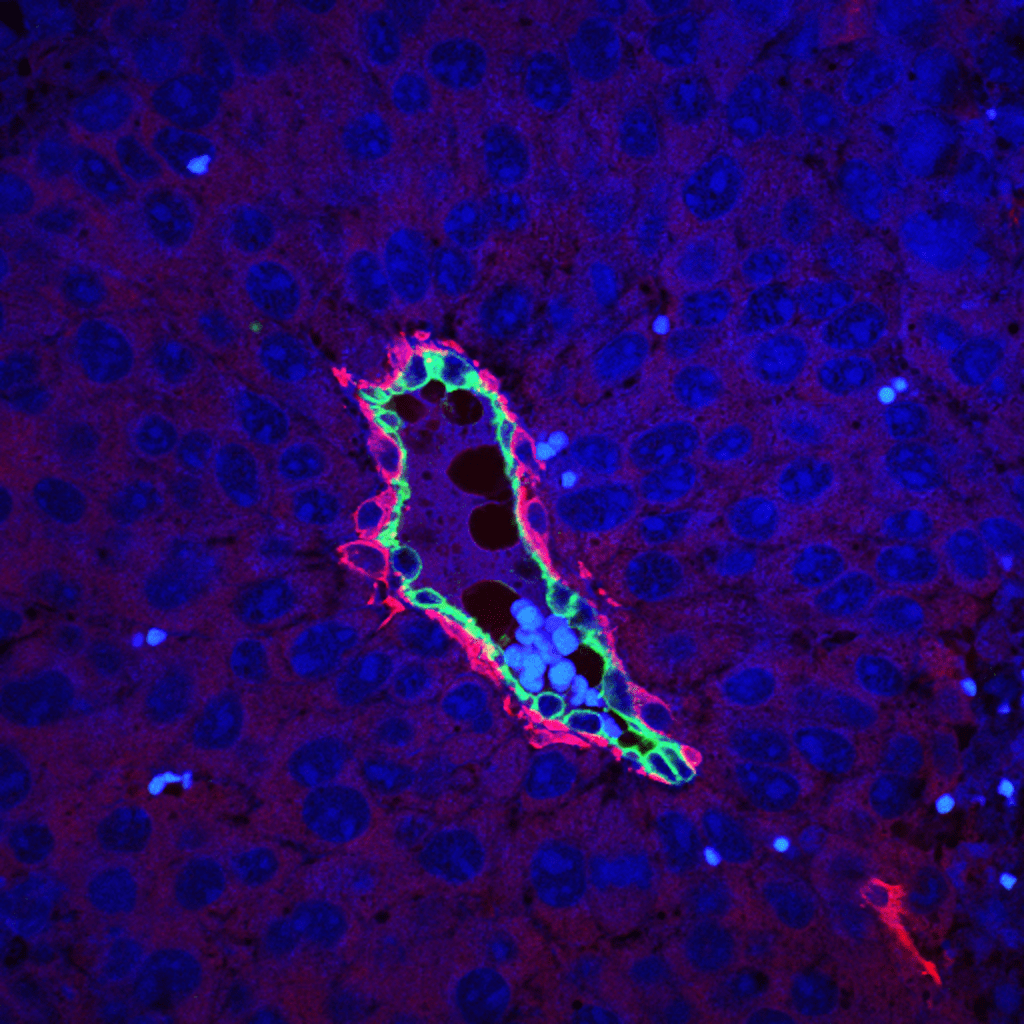
The study, published today in Cell and primarily funded by Cancer Research UK and Worldwide Cancer Research, describes a direct relationship between a group of cells in blood vessels - known as pericytes - and cancer cells. The team showed that if pericytes lack a single molecule called β3-intergrin, they start to secrete chemicals that encourage cancer cells to divide, increasing the growth of the tumour.
Tumour growth is controlled by interactions between cancer cells and the surrounding microenvironment, including blood vessels. Just as blood vessels deliver oxygen and nutrients to our organs, they also deliver oxygen and nutrients to tumours in the body, helping to feed the cancer and aid its growth and spread. Blood vessels have two main layers: An internal layer of fragile endothelial cells and an outer layer of ‘scaffold’ like mural cells known as pericytes. Pericytes are thought to help support the physical structure of blood vessels, preventing them from collapsing. Other functions of these specialised cells have been somewhat of a mystery, and scientists around the world are now starting to further explore the roles of pericytes in cancer.
This study was jointly led by Professor Ping Pui Wong of Sun Yat-Sen University in China, and Dr José Manuel Muñoz Félix and Professor Kairbaan Hodivala-Dilke from BCI’s Centre for Tumour Microenvironment. They said:
“We were surprised when our experiments started to reveal this new discovery. It was exciting because it is the first to show that pericyte β3-integrin can actually control how the whole cancer grows. This is important because if we can understand better what really helps cancer growth, and what stops cancer growth, we may be able to find new and improved ways to treat cancer.”
New targets for treatment
The study demonstrated that lack of pericyte β3-integrin was associated with increased growth of melanoma, breast cancer, lymphoma and mesothelioma in patients. Laboratory tests identified that the chemicals secreted by pericytes defective in β3-integrin trigger a cascade of molecular changes in tumour cells that make them divide and proliferate more. By identifying these molecular vulnerabilities, the team were able to devise strategies to slow down cancer growth in the laboratory.
The study also offers a possible explanation for why cancers grow more in some people than others. This information could be valuable in the clinic if oncologists were able to use a patient’s tumour blood vessel β3-integrin status to predict how their cancer is likely to grow. Although a lot more work will be necessary to test this finding, such knowledge may help to inform doctors on how best to treat a patient before their cancer spreads.
The study was performed in collaboration with the Institute of Cancer Research, the University of Edinburgh and the University of East Anglia.
Category: General News, Publications

No comments yet