Researchers identify novel target that could improve the safety of CAR T cell therapy for pancreatic cancer
 Researchers from Barts Cancer Institute (BCI), Queen Mary University of London, have identified a protein that may represent a novel therapeutic target for the treatment of pancreatic cancer. Using this protein as a target, the team successfully created a CAR T cell therapy - a type of immunotherapy - that killed pancreatic cancer cells in a pre-clinical model.
Researchers from Barts Cancer Institute (BCI), Queen Mary University of London, have identified a protein that may represent a novel therapeutic target for the treatment of pancreatic cancer. Using this protein as a target, the team successfully created a CAR T cell therapy - a type of immunotherapy - that killed pancreatic cancer cells in a pre-clinical model.
CAR T cell therapy is an immunotherapy that has shown great promise for the treatment of some blood cancers; however, the treatment of solid tumours using this therapy has proved very difficult. One barrier to success is toxicity in tissues other than the cancer because most of the proteins currently used to target CAR T cells to pancreatic cancer cells and other solid tumours are present in low levels on other normal tissues, leading to toxic side effects.
In this study, published today in Clinical Cancer Research and funded by the charity Pancreatic Cancer UK, the team identified a protein called CEACAM7 that may represent a safer treatment target for the development of therapies against pancreatic ductal adenocarcinoma (PDAC), the most common type of pancreatic cancer.
By using a specialised technique called immunostaining, the team examined a panel of human PDAC samples, and normal tissues for the presence of CEACAM7. A large subset of PDAC samples tested expressed CEACAM7, but the protein was undetectable in a panel of normal tissues including tonsil, lung, liver, and prostate, suggesting that CEACAM7 may be an ideal target for CAR T cell development against pancreatic cancer.
To determine the potential of CEACAM7 as a treatment target, the team developed CAR T cells targeted to CEACAM7 and applied these to PDAC cell lines as well as a preclinical model of PDAC. The CAR T cells effectively targeted the CEACAM7-expressing cells in PDAC cell cultures, and eliminated cancer cells in a late-stage preclinical model of PDAC.
BCI’s Professor John Marshall, who led the study, said:
“This is an exciting development. Finding that CEACAM7 allows us to kill pancreatic cancer cells specifically with CAR T cells while having no significant toxicity in non-tumour tissues, gives us hope that this strategy could be effective in the future. It is also possible that other types of immune-based therapies could be directed to CEACAM7 for the treatment of pancreatic cancer.”
Dr Deepak Raj, postdoctoral researcher at BCI and first author of the study, said:
“As CEACAM7 is a poorly studied protein thus far, we were excited to find that it appears a promising CAR T-cell target on pancreatic cancer. It would be important to assess a larger number of antibodies against CEACAM7, not only to generate and test a larger panel of CAR T cells that may have increased efficacy against pancreatic cancer, but also to more conclusively rule whether low levels of CEACAM7 are present in normal tissues.”
How does CAR T cell therapy work?
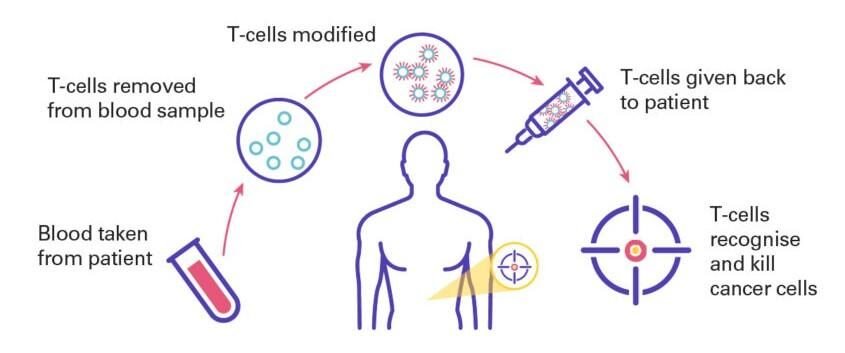
CAR T cell therapy utilises immune cells (called killer T cells) from the patient’s blood, which have a critical role in the immune response. Killer T cells are first isolated from the patient’s blood and modified in the laboratory to express special protein receptors on their surface, called Chimeric Antigen Receptors (CAR), creating CAR T cells. The CAR protein allows the CAR T cells to recognise a specific protein on the surface of cancer cells. CAR T cells are multiplied in the laboratory and then re-injected back into the patient where they recognise and kill cancer cells that have the target protein on their surface.
In this study, the team made a new CAR using part of an antibody to CEACAM7 from collaborator Professor Brad Nelson (British Columbia, Canada). They then modified killer T cells to present this new CAR protein on their surface that recognises and binds to CEACAM7, directing the killer T cells to kill only cells with CEACAM7, and these seem to be only pancreatic cancer cells.
Challenges in the treatment of pancreatic cancer
Pancreatic ductal adenocarcinoma (PDAC) is the most common type of pancreatic cancer and has the lowest survival rate of all the common cancers, as only about 7% of those diagnosed with this cancer type in the UK survive their cancer for 5 years or more. Diagnosis often comes too late due to a lack of definitive symptoms, by which point surgery to remove the tumour - which offers the greatest chance of a cure - is not possible. There is an urgent requirement for new and more effective targeted therapies.
Chris Macdonald, Head of Research at Pancreatic Cancer UK said:
“These findings are very encouraging and offer real hope that a new, innovative immunotherapy treatment for pancreatic cancer is on the horizon. For the first time a distinct and specific target protein for pancreatic cancer cells has been identified and, crucially, the brilliant team at Barts have shown that by focusing on it, they can destroy the cancer without damaging healthy tissue. This has never been done before in pancreatic cancer and marks an important step towards a desperately needed new treatment option, which could be both more effective and have fewer side-effects for patients.
Currently treatment options are limited and people affected by this devastating disease face incredibly low odds of survival. I look forward to seeing the results of targeting this protein in future clinical trials. I hope we’ll see these findings, along with the other research funded by Pancreatic Cancer UK’s Grand Challenge, benefit people with pancreatic cancer, the way we’ve seen new immunotherapy treatments benefit people with other types of cancer.”
Category: General News, Publications

Hemant Kocher 22/01/2021
Well done to Deepak. Exciting discovery which will help patients in the future.
Angela Bower 28/01/2021
Are you going to be carrying out clinical trials anytime soon and if so are you looking for volunteers? I was diagnosed with pancreatic cancer 6 months ago
Barts Cancer Institute 29/01/2021
While the team are excited by the data, the team are not yet in a position to take their CAR T cells into the clinic. They are looking for suitable pharmaceutical partners but this is likely to take some considerable time. We are sorry to give you this disappointing news but the team will keep trying to take this project forward.
Laura Comerford 12/04/2021
Hi I was wondering if you had made any progress with the clinical trial for the CAR T cells? I am looking for treatment options for my mum who was recently diagnosed with Pancreatic cancer.
Thank you for the amazing work you do.
Laura
Barts Cancer Institute 14/04/2021
The team are not yet in a position to take the CAR T cells into the clinic – this remains the team’s goal but it is likely to take some considerable time. We are sorry to give you this disappointing news, but the team will keep trying to take this project forward.
Richard T Soldan 04/05/2021
Are you aware of any medical facilities/schools in the USA that are looking for volunteers for clinical trials and studies? My wife was diagnosed in November 2020 and is young, healthy but dealing with encased arteries even after chemo treatments.
Barts Cancer Institute 07/05/2021
As we are an Institute based in the UK, we are not aware of the clinical trials currently recruiting in the USA. The following website is a database of clinical studies taking place around the world https://www.clinicaltrials.gov/
Susan Whittaker 06/05/2021
Hi.I too have been recently diagnosed with pancreatic cancer that metastasised to my liver. I too, like others are hoping for further research and hope for a better, longer future. This looks like a definite beginning. Pancreatic cancers seem to have been the poor relation as it were to press, so I hope that your search for a pharmaceutical partner is progressing positively. I am trying to keep going so that any new treatments that come forward I may be able to take advantage of. Us sufferers, however, don’t have the luxury of considerable time by the looks of it. Let’s hope there’s some more positive news shortly?
Laura Comerford 21/05/2021
Do you have any sort of time indicators for when the CAR T therapy might reach clinical trial stage? Is this more likely to be a period of months or years? Are there any other treatment options which we could look at for please?
Barts Cancer Institute 28/05/2021
Unfortunately, we are not able to provide a time indicator for this, nor are we in a position to advise on other treatment options – we are very sorry about this. The following website allows you to search for clinical studies taking place for pancreatic cancer https://www.clinicaltrials.gov/
anton 25/05/2021
My mother has recently been diagnosed with pancreatic cancer, She has a metastatic brain tumor as well, She is 65 years old, I’m afraid to lose her soon, we are eager to test new treatment methods on her disease.
Is there any update about this new method?
Barts Cancer Institute 28/05/2021
We are very sorry to hear of your mother’s diagnosis. Unfortunately, we are not able to provide an update on this research at this time, but the team will keep trying to take this project forward.
Nicola 01/10/2021
Is there any update on this, have you been able to secure a pharmaceutical partner?
Barts Cancer Institute 05/10/2021
We’re very sorry but unfortunately we do not have an update on this research at this time
Tina Machell 10/10/2021
My sister has been diagnosed with stage 4 pancreatic cancer with a prognosis of 5-7 months life expectancy . She is a strong and fit and just turned 60
Please can she be included in this trial
Barts Cancer Institute 13/10/2021
We are very sorry to hear of your sister’s diagnosis. Unfortunately, the team are not yet in a position to take the CAR T cells into the clinic and they are still working to identify a suitable pharmaceutical partner. We are sorry to give you this disappointing news.
Nicola 30/11/2021
Are you still trying to find a pharmaceutical partner and is this the only barrier to human trials or is this on hold? Thanks
Barts Cancer Institute 08/12/2021
Thank you for your comment. The team is still working to secure a pharmaceutical partner to take this work forward.
Lindsey 27/01/2022
When you get the go ahead for clinical trials where will this be published please and how do people register for this? This work is amazing and offering a glimmer of hope for people diagnosed with this.
Barts Cancer Institute 03/02/2022
Thank you for your comment. This research is not yet at the stage of reaching a clinical trial, and the team are still working to secure a suitable pharmaceutical partner to take this project forward. Information about clinical trials is made publicly available at https://clinicaltrials.gov/, including information on eligibility criteria and how to join the trial.
Suzanne 18/10/2022
Hello, my sister has been diagnosed with pancreatic cancer and is suffering g a lot from the affects of the chemotherapy. Are you any further along with partnering with a pharmaceutical company for your CAR T immunotherapy work? My sister would very much want to take part in a clinical trial for it. Please let me know how close you are to beginning the treatment. Thank you so much.
Barts Cancer Institute 21/10/2022
We are very sorry to hear of your sister’s diagnosis. Unfortunately, we do not have any updates to provide at this stage.
Abdulateef Akinwande 27/10/2022
I’ll like to know more about this research, Is there any way I can be receiving emails about this research?
Barts Cancer Institute 07/11/2022
Thank you for getting in touch. There is currently no set up for receiving email updates about this research, apologies for this. Any significant updates about this research will be communicated via the news section of our website.
Abir aftab 31/10/2022
Hi I have been diagnosed with pancreatic cancer and wondering how I can going this or If any progress on offering this to people has been made ?
Barts Cancer Institute 07/11/2022
We are very sorry to hear of your diagnosis. This research is not yet at the stage of reaching a clinical trial, and we do not have any updates to provide at this stage. We are sorry to give you this disappointing news.